Supported by Dr. Osamu Ogasawara and  providing providing  . . |
|
Last data update: 2014.03.03 |
Spatial bias detectionDescriptionThis function detects spatial bias on array CGH. Usage## S3 method for class 'arrayCGH' detectSB(arrayCGH, variable, proportionup=0.25, proportiondown,type="up", thresholdup=0.2, thresholddown=0.2, ... ) Arguments
DetailsYou must run the ValueAn object of class
NotePeople interested in tools for array-CGH analysis can visit our web-page: http://bioinfo.curie.fr. Author(s)Philippe Hupé, Philippe.Hupe@.curie.fr. ReferencesP. Neuvial, P. Hupé, I. Brito, S. Liva, E. Manié, C. Brennetot, A. Aurias, F. Radvanyi, and E. Barillot. Spatial normalization of array-CGH data. BMC Bioinformatics, 7(1):264. May 2006. See Also
Examples
data(spatial) ## arrays with local spatial effects
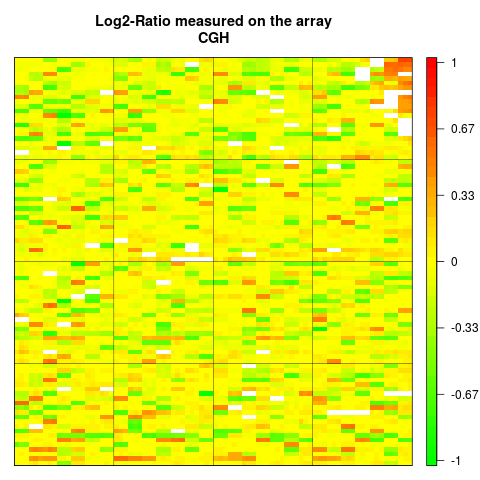
## Plot of LogRatio measured on the array CGH
arrayPlot(edge,"LogRatio", main="Log2-Ratio measured on the array
CGH", zlim=c(-1,1), bar="v", mediancenter=TRUE)
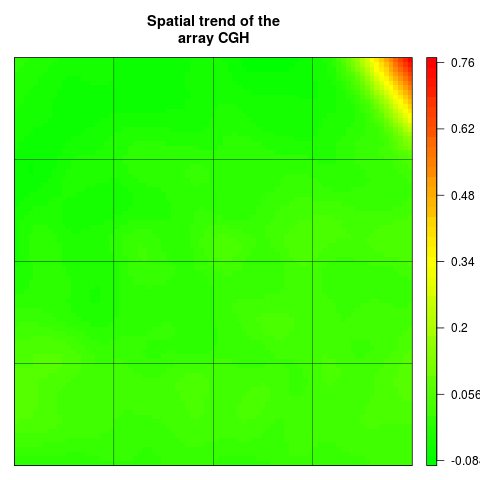
## Spatial trend of the scaled log-ratios (the variable "ScaledLogRatio"
## equals to the log-ratio minus the median value of the corresponding
## chromosome arm)
edgeTrend <- arrayTrend(edge, variable="ScaledLogRatio",
span=0.03, degree=1, iterations=3, family="symmetric")
arrayPlot(edgeTrend, variable="Trend", main="Spatial trend of the
array CGH", bar="v")
## Not run:
## Classification with spatial constraint of the spatial trend
edgeNem <- nem(edgeTrend, variable="Trend")
arrayPlot(edgeNem, variable="ZoneNem", main="Spatial zones identified
by nem", bar="v")
# Detection of spatial bias
edgeDet <- detectSB(edgeNem, variable="LogRatio", proportionup=0.25,type="up", thresholdup=0.15)
arrayPlot(edgeDet, variable="SB", main="Zone of spatial bias in red", bar="v")
# CGH profile
plot(LogRatio ~ PosOrder, data=edgeDet$arrayValues,
col=c("black","red")[as.factor(SB)], pch=20, main="CGH profile: spots
located in spatial bias are in red")
## End(Not run)
Results
R version 3.3.1 (2016-06-21) -- "Bug in Your Hair"
Copyright (C) 2016 The R Foundation for Statistical Computing
Platform: x86_64-pc-linux-gnu (64-bit)
R is free software and comes with ABSOLUTELY NO WARRANTY.
You are welcome to redistribute it under certain conditions.
Type 'license()' or 'licence()' for distribution details.
R is a collaborative project with many contributors.
Type 'contributors()' for more information and
'citation()' on how to cite R or R packages in publications.
Type 'demo()' for some demos, 'help()' for on-line help, or
'help.start()' for an HTML browser interface to help.
Type 'q()' to quit R.
> library(MANOR)
Loading required package: GLAD
######################################################################################
Have fun with GLAD
For smoothing it is possible to use either
the AWS algorithm (Polzehl and Spokoiny, 2002,
or the HaarSeg algorithm (Ben-Yaacov and Eldar, Bioinformatics, 2008,
If you use the package with AWS, please cite:
Hupe et al. (Bioinformatics, 2004, and Polzehl and Spokoiny (2002,
If you use the package with HaarSeg, please cite:
Hupe et al. (Bioinformatics, 2004, and (Ben-Yaacov and Eldar, Bioinformatics, 2008,
For fast computation it is recommanded to use
the daglad function with smoothfunc=haarseg
######################################################################################
New options are available in daglad: see help for details.
Attaching package: 'MANOR'
The following object is masked from 'package:base':
norm
> png(filename="/home/ddbj/snapshot/RGM3/R_BC/result/MANOR/detectSB.Rd_%03d_medium.png", width=480, height=480)
> ### Name: detectSB
> ### Title: Spatial bias detection
> ### Aliases: detectSB detectSB.default detectSB.arrayCGH
> ### Keywords: models spatial
>
> ### ** Examples
>
> data(spatial) ## arrays with local spatial effects
>
> ## Plot of LogRatio measured on the array CGH
> arrayPlot(edge,"LogRatio", main="Log2-Ratio measured on the array
+ CGH", zlim=c(-1,1), bar="v", mediancenter=TRUE)
>
> ## Spatial trend of the scaled log-ratios (the variable "ScaledLogRatio"
> ## equals to the log-ratio minus the median value of the corresponding
> ## chromosome arm)
>
> edgeTrend <- arrayTrend(edge, variable="ScaledLogRatio",
+ span=0.03, degree=1, iterations=3, family="symmetric")
> arrayPlot(edgeTrend, variable="Trend", main="Spatial trend of the
+ array CGH", bar="v")
>
> ## Not run:
> ##D ## Classification with spatial constraint of the spatial trend
> ##D edgeNem <- nem(edgeTrend, variable="Trend")
> ##D arrayPlot(edgeNem, variable="ZoneNem", main="Spatial zones identified
> ##D by nem", bar="v")
> ##D
> ##D # Detection of spatial bias
> ##D edgeDet <- detectSB(edgeNem, variable="LogRatio", proportionup=0.25,type="up", thresholdup=0.15)
> ##D arrayPlot(edgeDet, variable="SB", main="Zone of spatial bias in red", bar="v")
> ##D
> ##D # CGH profile
> ##D plot(LogRatio ~ PosOrder, data=edgeDet$arrayValues,
> ##D col=c("black","red")[as.factor(SB)], pch=20, main="CGH profile: spots
> ##D located in spatial bias are in red")
> ## End(Not run)
>
>
>
>
>
> dev.off()
null device
1
>
|